

HK L-137 increases the production of Th1-type cytokines from macrophage and dendritic cells. Th1-type immune responses, which are regulated by these cytokines, are responsible for the protection against virus infection. Since HK L-137 enhances the Th1-type immune responses, it is expected that HK L-137 exerts a protective effect against virus infection.
HK L-137 was orally administered to mice, and the effect on influenza virus infection was examined. In the HK L-137 group, there was a significant secretion of IFN-β, which is one of the cytokines that play an extremely important role in the host defense against infection with viruses and bacteria. In addition, HK L-137 prolonged the survival period of mice that were infected with highly virulent strains of the influenza virus.
A lethal dose of a highly virulent strain of influenza virus(100 pfu) was nasally infected to mice, and the serum concentration of IFN-β, the virus titer in pulmonary tissues, and the survival period were examined. Mice in the HK L-137 group were orally administered 75 mg/kg of HK L-137 once daily for 15 days from 7 days before the infection.
In the control group, IFN-β was not detected in the serum, but high levels of IFN-β were detected in the HK L-137 group. The virus titer in the pulmonary tissues decreased drastically and the survival period was prolonged in the HK L-137 group.
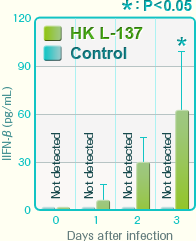
Serum concentration of IFN-β after influenza virus infectionIFN-β was not detected in the control group, but high levels of IFN- IFN-β were detected in the mice orally administered HK L-137.
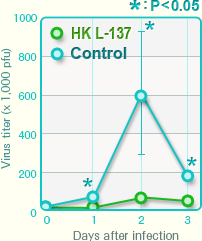
Influenza virus titer in pulmonary tissues
Administration of HK L-137 significantly suppressed an increase in the influenza virus titer.
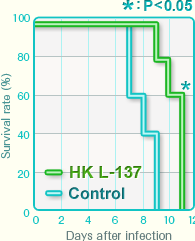
Survival period after influenza virus infection
Oral administration on HK L-137 prolonged the survival period after viral infection.
Oral administration of HK L-137 significantly suppresses the growth of influenza virus in infected mice, which may lead to prolongation of the survival period of the mice. It is possible that HK L-137 suppresses the virus growth by inducing IFN-β production at the early stages of infection.
A wide variety of viruses can effectively spread through their suppression of IFN production. HK L-137 induced IFN-β production even in the mice infected with virulent influenza viruses that force the host not to produce IFN-β. Therefore, HK L-137 will be effective to prevent virus infection, especially at the early stage of the infection.
Reference:Int Immunopharmacol 9: 1122-1125, (2009)